It’s here: Levetiracetam is the first ever 3D printed drug to be approved by the United States Food and Drug Administration (FDA). Levetiracetam is used to treat convulsions and is prescribed to patients with epilepsy and has been distributed as Keppra® since 2008. Aprecia Pharmaceuticals has developed a new version of Levetiracetam, which they’ve dubbed SPRITAM®, that quick dissolves in a small drink of liquid. Aprecia accomplished this by using 3D printing on their own ZipDose® platform.
By combining 3DP technology with a highly-prescribed epilepsy treatment, SPRITAM is designed to fill a need for patients who struggle with their current medication experience… This is the first in a line of central nervous system products Aprecia plans to introduce as part of our commitment to transform the way patients experience taking medication.”
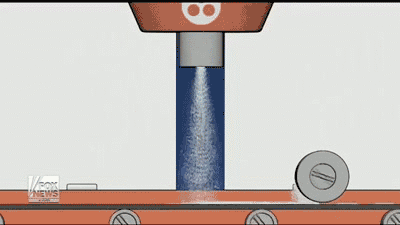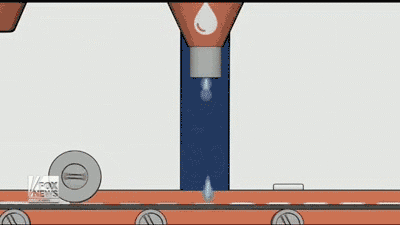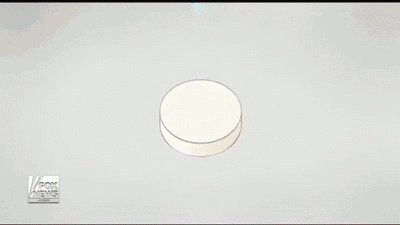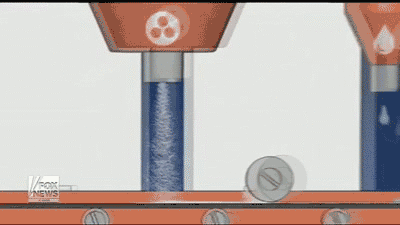
By using a 3D printer, Aprecia is able to lay down layers of the drug needed and make the pills more concentrated, it also dissolves quicker with a small drink of water than normal pills. Aprecia is also able to make a higher dose in a smaller pill, eliminating the need for patients to swallow a handful of pills, which helps those who have trouble with pills. In the GIF below you can see how the process is done.
In my experience, patients and caregivers often have difficulty following a treatment regimen. Whether they are dealing with a swallowing disorder or the daily struggle of getting a child to take his or her medication, adherence can be a challenge.”
Being able to layer the medication and produce higher doses will also help those who may have to take their medication multiple times a day by letting them take only one pill a day.
What do you think of SPRITAM®? Let us know in the comments below or on Google+, Facebook and Twitter.
[button link=”http://www.news-medical.net/news/20150805/First-drug-produced-by-3D-printing-is-approved-by-FDA.aspx” icon=”fa-external-link” side=”left” target=”blank” color=”285b5e” textcolor=”ffffff”]Source: News-Medical[/button]Last Updated on November 27, 2018.