Let’s face it, we’re a planet of power consuming beings and more power is the cry of every mobile device owner both power and regular user. You can’t go through the day without seeing dozens of people wall hugging with their respective devices tethered to an outlet somewhere. While smartphone technology has grown rapidly, battery technology just hasn’t kept up at the same pace. I suppose one could argue that users may be demanding just too much from their devices as well. Either way, new battery tech is on the horizon in the form of lithium metal batteries and a research team at Cornell
may have moved it one step closer to reality.
The problem with lithium metal batteries is that the lithium in the anode is so reactive that, when you recharge the battery, it will form dendrites (or tree-like bumps) that eventually extend through the electrolyte all the way to the cathode, effectively causing a short-circuit which can lead to thermal runaway and some pretty scary safety hazards.
Other researchers have attempted to use ceramic membranes to deter the growth of dendrites but that also decreases performance and increases heat, both not acceptable conditions.
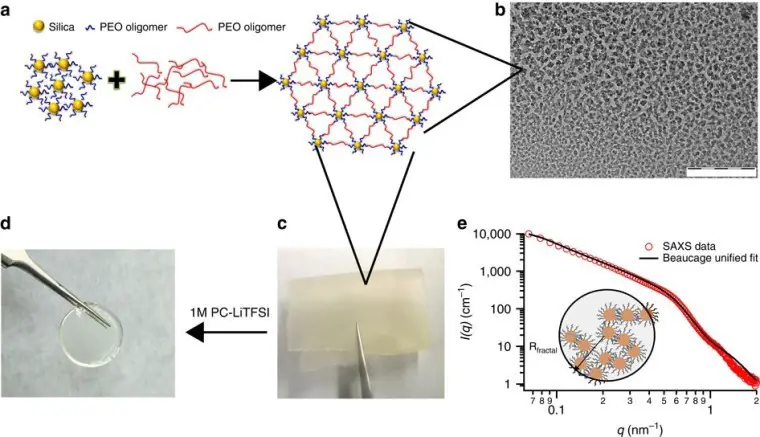
Chemical and biomolecular engineering professor Lynden Archer’s team has created a new membrane “by grafting polyethylene oxide onto silica to form nanoscale organic hybrid materials” – “hairy” nanoparticles that can effectively stop dendrite growth at room temperature.
The researchers claim that this solution does not require a radical redesign in battery technology, and can be “as effective as batteries based on other metals, such as sodium and aluminum, that are more earth-abundant and less expensive than lithium and also limited by dendrites.”
All the technical parameters behind the research can be found in the published paper, there is more than enough there to occupy your day. The bottom line is, Cornell and other researchers are working hard to make battery power more efficient and hopefully lithium metal batteries are not too far away.
What do you think of lithium metal batteries and this research from Cornell? Let us know in the comments below or on Google+, Facebook and Twitter.
[button link=”http://www.neowin.net/news/cornell-team-brings-lithium-metal-battery-one-step-closer-to-your-digital-devices” icon=”fa-external-link” side=”left” target=”blank” color=”285b5e” textcolor=”ffffff”]Source: Neowin[/button]Last Updated on January 23, 2017.










