
After recent discoveries of Earth-like planets and all manner of strangeness in our stellar neighborhood, possibly our next question should be: How do we look for alien life? Many say the tell-tale signs of life are carbon and probably water. Seems reasonable, all life on Earth essentially consists of carbon compounds suspended in water after all. However, look a little deeper and you’ll find there is a more subtle candidate in our quest for life and it is, surprisingly enough, uranium!
For ten years, geophysicist J. Marvin Herndon has presented increasingly persuasive arguments that at the very center of the Earth, within the inner core, there exists a five mile in diameter sphere of uranium which acts as a natural nuclear reactor.
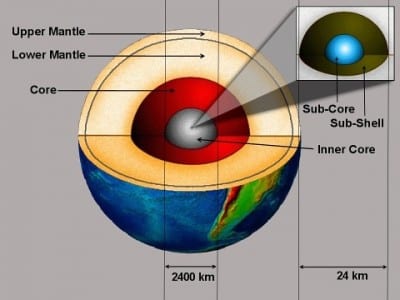 Dr. Herndon likes to term this beast the “georeactor.”
Dr. Herndon likes to term this beast the “georeactor.”
This four terawatt georeactor is likely to have powered the formation of our atmosphere and oceans, around a billion years after the Earth was born. Today it continues to protect us by generating a magnetosphere, a magnetic field which surrounds our planet that shields us from the sun’s hard radiation. Nice as it may seem, our close neighbor the sun is effectively a huge unshielded fusion reactor. Fortunately too, our magnetosphere also protects Earth’s rich atmosphere, stopping it from being eroded into space by solar radiation. For comparison, Mars lost its magnetosphere early on in its life and after a few billion years of solar irradiation – “pretty” would be a euphemism…
Before even the planets were formed uranium also likely supplied the initial burst of energy required to ‘ignite’ our sun’s fusion reaction, similar to the way uranium is used to initiate a fusion bomb. There are plenty of potential suns out there which accumulated sufficient mass for fusion but then failed to ignite, choosing instead to become cold (and lonely) brown dwarfs.
So where you find uranium you could also find young virile suns, with many “lively” planets as consorts. How Zen, that an element like uranium, which seems so invidious to life, might prove so essential.
Last Updated on November 27, 2018.











Comments are closed.